Scanning force microscopy
We conduct regular training courses on atomic force microscopy and spectroscopy. We also have instruments within a biosafety level 2 laboratory, where access is restricted. Contact us and we will find a mode and the right atomic force microscope or spectroscope for your requirements. Depending on the type of samples and the number of experiment series, you will be trained or we will carry out the measurements for you. Members of the UdS can find further information in the UdS Instrumentation Database in the intranet.
If you have any questions, please contact Dr. Hendrik Hähl.
JPK NanoWizard 4 XP

Atomic force microscope and force spectroscope optimized for characterizing the adhesion of cells, describing the elasticity of soft surfaces, or determining the adhesion force of micrometer- to nanometer-sized objects (e.g., colloids, bacteria, small, possibly coated glass or polymer spheres). A top down microscope is attached for observation as well as an inverted fluorescence microscope, which also offers confocal imaging. The instrument is located in a biosafety level 2 laboratory.
Features
NanoWizard 4XP
- Scanning in air/gases/fluids
- Scanning modes: QI™ mode, contact mode with lateral force, advanced AC modes, electrical & magnetic imaging mode options
- Maximum scan range (XxYxZ): 100x100x15 µm³
- Fast scanning option (150 Hz (lines/second) for scan ranges up to 2 µm)
- Cytosurge FluidFM® microfluid control system
- Alternative CellHesion®200 scan head with 100 µm z-range
Inverted Microscope
- Zeiss Axio Observer 7
- LSM 900
- LED fluorescent light source Kolibri 7
Bruker FastScan Bio

Operating Modes:
- ScanAsyst in Air/Fluid
- Tapping Mode in Air/Fluid
- Contact Mode in Air/Fluid
- Lateral Force Microscopy
- Nanomechanical Mapping (PeakForce QNM)
- Force Volume
- Electrical & Magnetic Lift Modes
Sample Specifications:
- Max. diameter ca. 20 cm, thickness: max. 1.2 cm
- Maximum scan range: 90 μm x 90 µm in air, 30 µm x 30 µm in liquid
- Maximum scan height: ca. 3 µm
Applications:
We use highly compressed hydroxyapatite pellets as a first order model system for the mineral phase of tooth enamel. These pellets, when properly prepared, feature locally a very low surface roughness below 1 nm (rms). Upon etching (e.g., as a part of a cleaning procedure), domains become visible due to different etching rates of differently oriented crystallites.

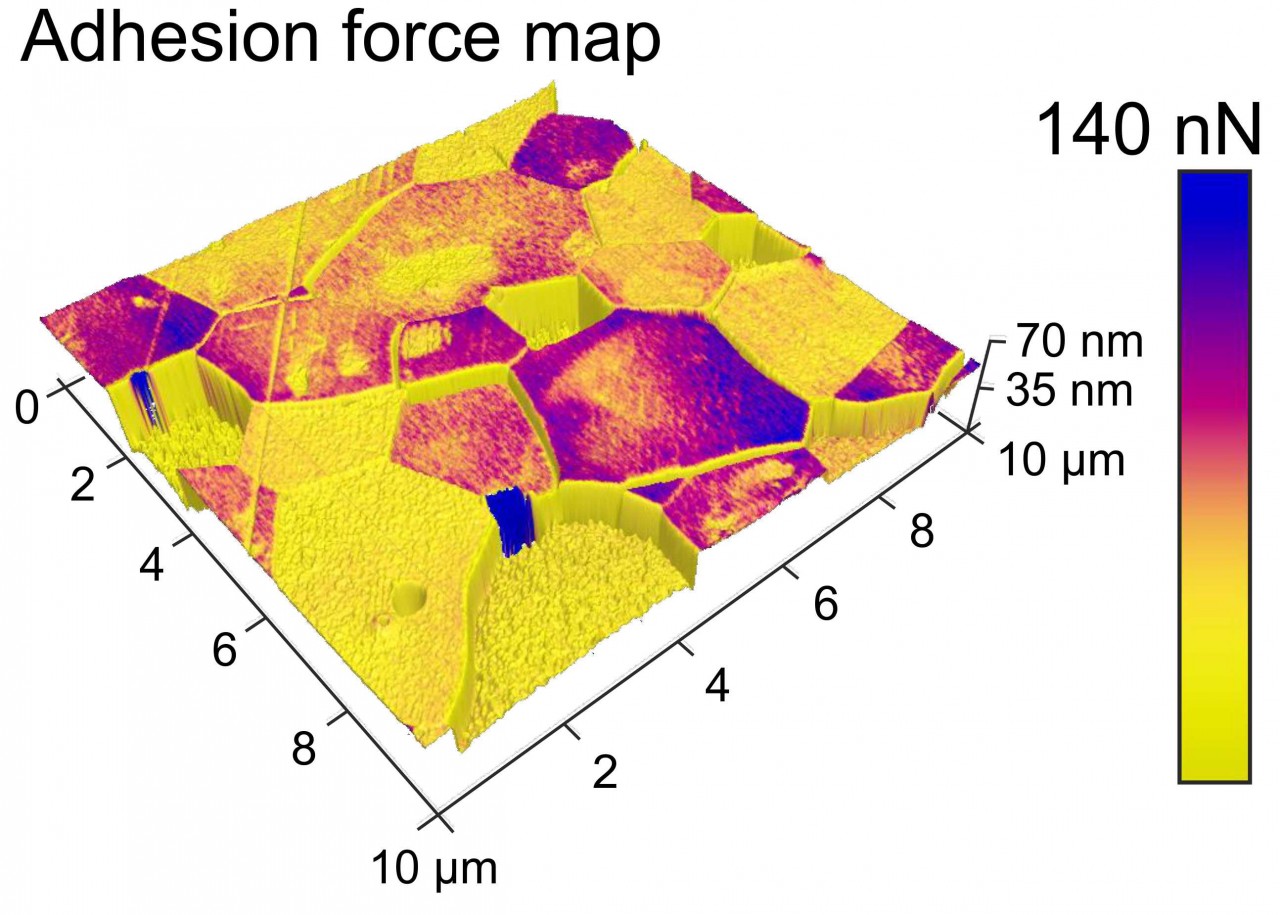
With the FastScan Bio, we are also able to map mechanical surface properties or adhesion forces. Adhesion force maps of hydroxyapatite surfaces reveal different forces on different grains. However, the origin of these differences is yet unknown and its exploration is part of our ongoing research.

 Deutsche Version
Deutsche Version